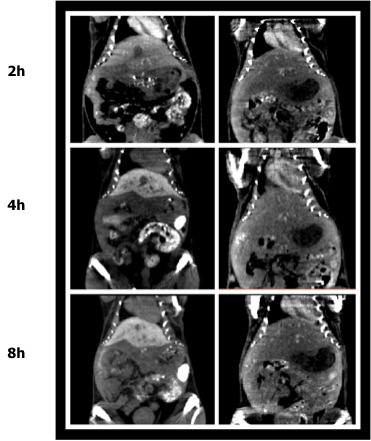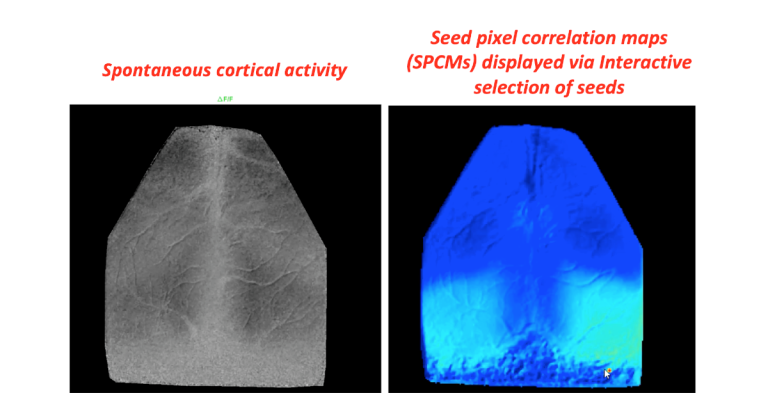
How spontaneous is spontaneous neural activity?
In their excellent review article titled ‘Not so spontaneous: Multi-dimensional representations of behaviors and context in sensory areas’ (ref. https://www.cell.com/neuron/pdf/S0896-6273(22)00588-8.pdf), Avitan and Stringer propose that